ZERO CONFLICTS OF INTEREST.
Just Compliance: Why a truly Independent IRB Matters.
In clinical research, ethical oversight isn’t just important—it’s paramount. Institutional Review Boards (IRBs) serve a critical role in safeguarding human research participants and ensuring compliance with regulatory standards. But what happens when an IRB has financial interests that extend beyond its core ethical review mission?
Over the last several years, many of the largest IRBs have focused on expanded services outside of ethical review, in areas such as: site selection, feasibility, participant recruitment, technology integration, and research staffing—creating the potential for a troubling conflict of interest. Can an IRB truly remain impartial when it profits from the very studies it reviews? Increased services and growing focus away from ethical review may increase the risk of influencing ethical oversight, the integrity of research and the safety of participants.

The Problem with Conflicted IRBs
When an IRB is owned by or affiliated with a company providing other research-related services, a potential conflict of interest may arise. This potential conflict is important to be aware of, as trust within research relies both in the actions of those involved with overseeing the research, as well as the participant perspective of trust from those involved.
Some participants may have issues with trusting an IRB’s independence when the same company is profiting from so many other services associated with the trial.
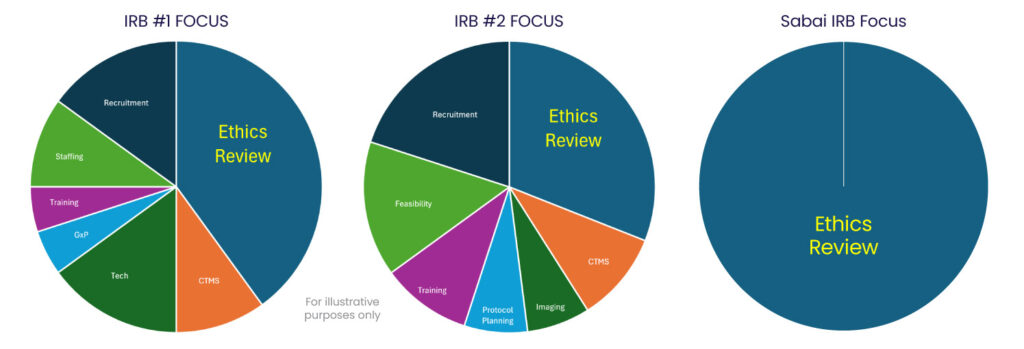
Scientific integrity and participant protection demand clear separation of roles in research. Federal regulations and accrediting bodies stress the need for IRBs to operate free from conflicts of interest, a safeguard born from past ethical failures. The International Council for Harmonisation of Good Clinical Practice (ICH GCP E6(R2) and E6(R3)) reinforces the necessity of independent IRB oversight, sponsor accountability, and investigator responsibility. When these roles blur, ethical obligations may be compromised, eroding trust in research.
A truly independent IRB isn’t just a requirement—it’s the foundation of ethical research. Regulators enforce these boundaries to prevent undue influence and protect participants.
At Sabai, we do things differently. 100% Independent. 100% Ethical. 100% focused on IRB and IBC Review.
Sabai is committed to one thing: ethical and regulatory review services. We do not bundle our review solutions with technology and ancillary services. Our sole focus is ensuring that studies meet the highest ethical and regulatory standards, without external business pressures influencing our decisions.
We have ZERO Conflicts of Interest.
Why Independence Matters
- Uncompromised Ethics: Our decisions are based on science, ethics and compliance. Not business relationships.
- Regulatory Trust: Sponsors, investigators and regulatory agencies can be confident that our reviews are conducted without outside influence.
- Participant Protection: With no competing interests, our IRB remains dedicated to the safety and well-being of research participants.
Choose Transparency. Choose Integrity. Choose Sabai.
If you’re looking for an IRB that prioritizes ethics, Sabai is your trusted partner. Contact us today to learn more about how our independent IRB services can support your research with the highest level of integrity.
WHAT SETS US APART
What Our Clients Say
"Christine was very patient with our questions and did a great job explaining and helping the site learn—[made] the process incredibly seamless and we look forward to our next project together."
"Many thanks for your excellent service. [You’ve] been the easiest and most collaborative and communicative IRB to work with. Hoping to cross paths again!"
"The best vendor I have ever worked with."
"We love you guys."
"Accelerated our timelines and beat our projections for our sponsor, with the added complexity of IBC on top of IRB regulatory."
"Every time I interact with your team I walk away with a smile. For Shield, Heidi has been my lifesaver: pragmatic, understanding, and action-oriented. I have come to her feeling overwhelmed and unsure of how to tackle complex issues, and within the span of a single call she develops a plan and I am able to walk away with 1000 pounds lifted off my shoulder."


